Published by: Nuru
Published date: 26 Jun 2021
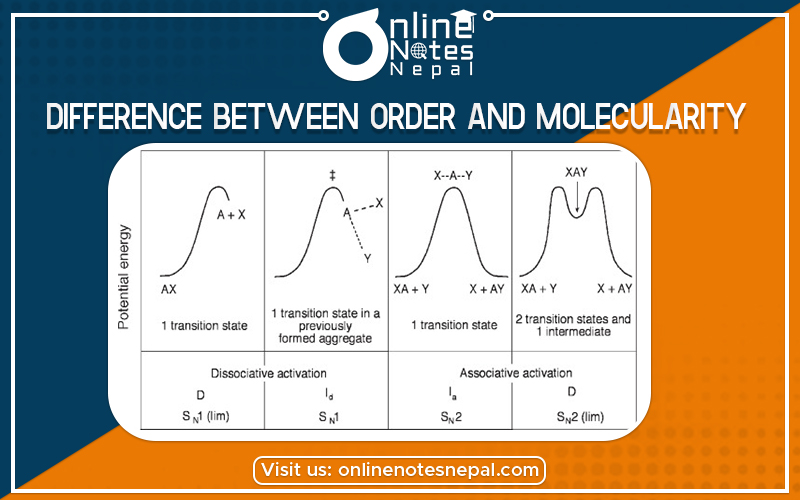
The difference Between Order And Molecularity of Reaction is explained below:
The order of reaction can be defined as the power dependence of rate on the concentration of all reactants. For example, the rate of a first-order reaction is dependent solely on the concentration of one species in the reaction.
The molecularity of a reaction refers to the number of atoms, molecules, or ions that must undergo a collision with each other in a short time interval for the chemical reaction to proceed.
Order of reaction |
Molecularity of reaction |
| 1) The actual concentration of reactants that affects the rate of a chemical reaction is called an order of a reaction.2) It is an experimental parameter. 3) It is calculated by the sum of the power of the reactant in a given rate law equation. 4) Its value can be 0. |
1) The total number of reactant molecules involved in a chemical reaction by effective collision to form products under suitable temperature & pressure. 2) It is a theoretical parameter. 3) It can be calculated by the sum of the coefficient of the reactant in the rate law equation. 4) Its value can’t be 0. |